Port wine stains treatment12/11/2023 Uncontrolled or non-comparative studies (essentially case series) encompassed one and the same treatment in all patients. In case of disagreement between the two reviewers, a third reviewer (CvdH) was consulted until consensus was reached.Ĭharacteristics of the outcome measure, measurement instrument, scoring system, interventions, study design and population, assessors, blinding of outcome assessment, statistical analysis, follow-up duration, the reporting of results, country of the first author, and the registration of adverse events were extracted independently by two investigators (IvR, SC). Titles, abstracts, full text versions of selected studies, and reference lists of included studies were screened independently by two investigators (IvR, SC) using Rayyan. We found that the outcomes in small case series (less than five PWS patients), letters, conference abstracts, short reports, and study protocols in international trial registries were, in general, described with insufficient detail. Studies that exclusively included patients with syndromic forms of PWS (e.g., Sturge-Weber syndrome, Klippel-Trénaunay syndrome, etc.) were excluded. Clinical efficacy assessment was defined as a visual evaluation of appearance or improvement without the aid of an (objective) instrument. The results of this study could help to inform the future development of a standardized clinical scoring system for PWS and to improve the quality of PWS research.įull-text, original studies with a therapeutic intervention performed in PWS patients and with a form of clinician-, observer-, or patient-reported clinical efficacy assessment as study outcome were included. Finally, the methodological quality of included studies was investigated using the Downs & Black risk of bias checklist. Additionally, the use of other outcome measures in these studies was assessed. Therefore, the primary goal of this study was to systematically map and analyze the use of clinical PWS outcome measures (i.e., an observer-, clinician-, or patient-reported visual assessment of treatment efficacy) in prospective PWS studies since 2005. Nevertheless, few studies have devoted attention to PWS outcome measures and no study has systematically analyzed the use of PWS outcome measures. The outcome measure heterogeneity has also precluded data syntheses in previous PWS meta-analyses. This review was limited in its scope because of the large variety of clinical scoring systems. Recently we have performed an analysis of clinical outcomes in PWS trials published since 1986. Increased awareness of the importance of (high-quality) outcome measures has led to a rise in outcome measure research, especially in dermatology (in particular for psoriasis, atopic dermatitis, and more recently vitiligo ). Moreover, standardization of outcome measures is imperative for enabling comparisons between studies. Valid, relevant, and reliable outcome measures are required to accurately gauge treatment effects and compare treatment protocols and therapeutic interventions. 
Despite therapeutic developments, complete PWS resolution remains rare. PWS are most frequently located in the face and neck and can cause functional impairment, skin and soft tissue hypertrophy, and glaucoma, as well as substantial psychosocial morbidity. 
Lesions initially present as flat, red-to-pink patches and darken and thicken with age. Learn more about Amazon Lockers.Port wine stains (PWS) are congenital vascular malformations that occur in approximately 0.3–0.9% of infants. 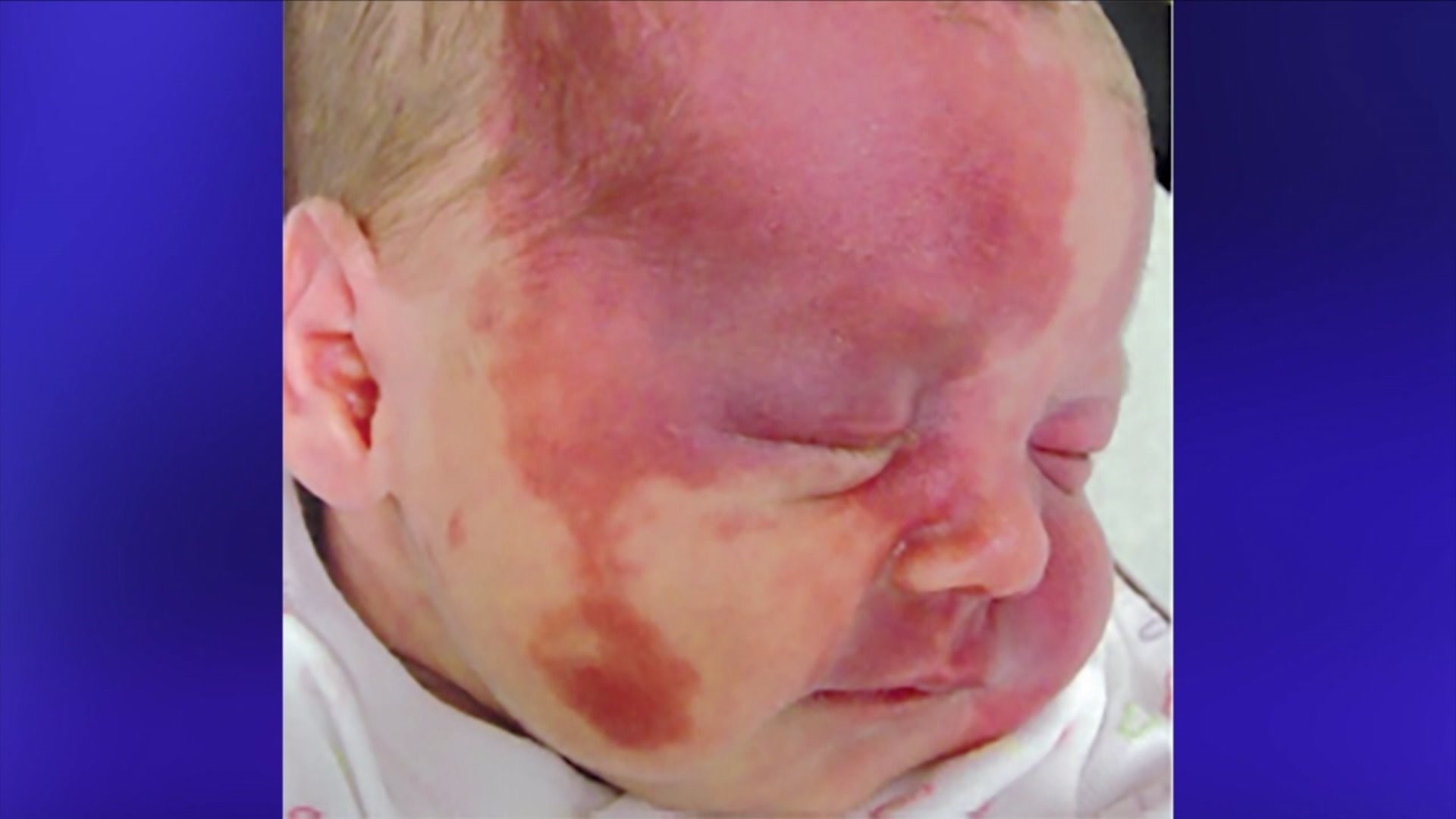 
Children’s Health is proud to become the first pediatric health system in the country to offer Amazon Lockers, self-service kiosks that allow you to pick up your Amazon packages when and where you need them most – 24 hours a day, seven days a week.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
